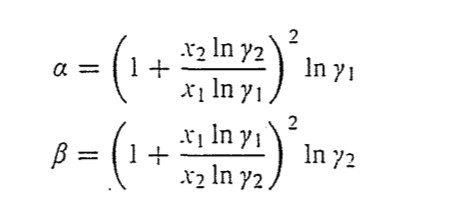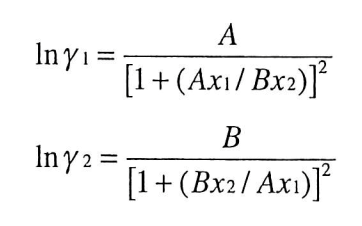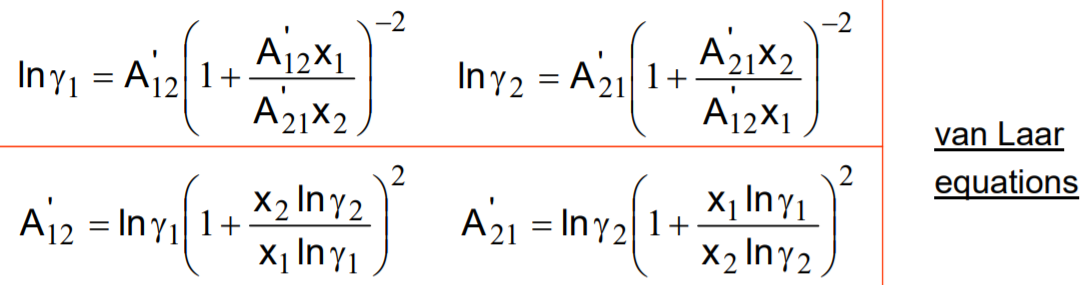![PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0b4e610592ffe5443a53149e7d185fe8b3348cb2/5-Table3-1.png)
PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar
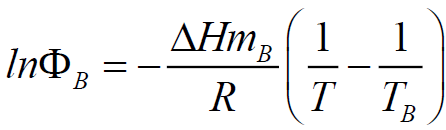
Eutectic Mixture of Bismaleimides Containing Polymethylene Flexible and Aramide-Arylate Mesogen Groups
![PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0b4e610592ffe5443a53149e7d185fe8b3348cb2/4-Table1-1.png)
PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar
DETERMINATION OF COMPOSITION-DEPENDENI' LIQUID ACTIVITY COEFFICIENrS1 BY USE OF THE VAN LAAR EQUATION GARRY LEONARD PIGG MASTER
![SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat](https://cdn.numerade.com/ask_images/1604588eda0f4ffc8897da2c6b3d4fee.jpg)
SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat
DETERMINATION OF COMPOSITION-DEPENDENI' LIQUID ACTIVITY COEFFICIENrS1 BY USE OF THE VAN LAAR EQUATION GARRY LEONARD PIGG MASTER