
Pancake Bonding: An Unusual Pi‐Stacking Interaction - Kertesz - 2019 - Chemistry – A European Journal - Wiley Online Library
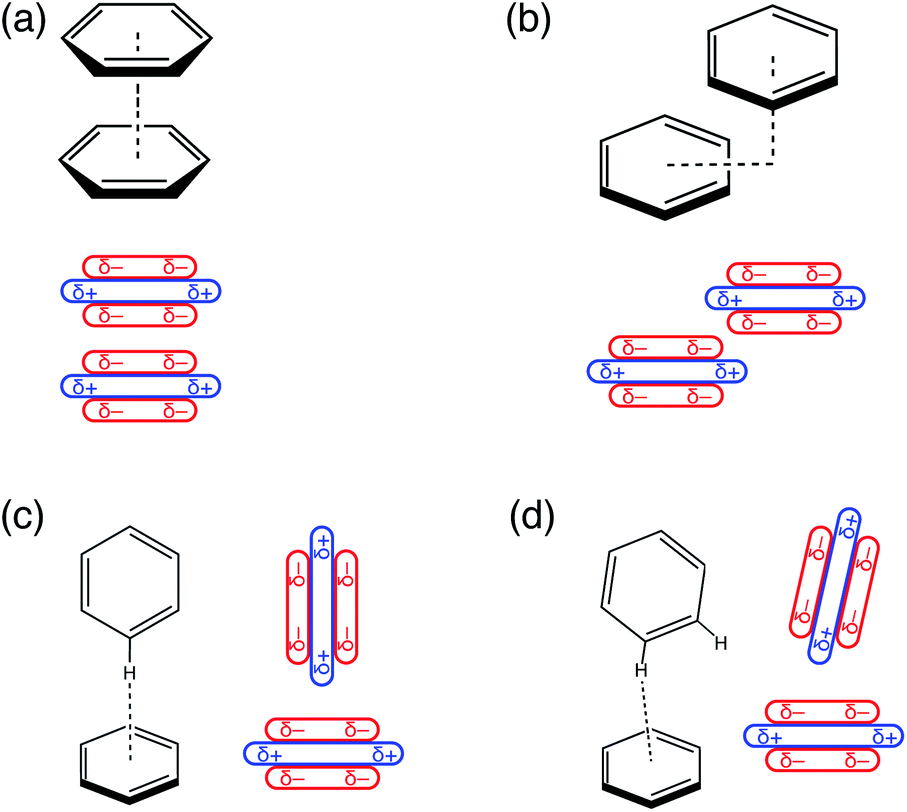
Electrostatics does not dictate the slip-stacked arrangement of aromatic π–π interactions - Chemical Science (RSC Publishing) DOI:10.1039/D0SC02667K

π–π Stacking Interaction in an Oxidized CuII–Salen Complex with a Side‐Chain Indole Ring: An Approach to the Function of the Tryptophan in the Active Site of Galactose Oxidase - Oshita - 2019 -
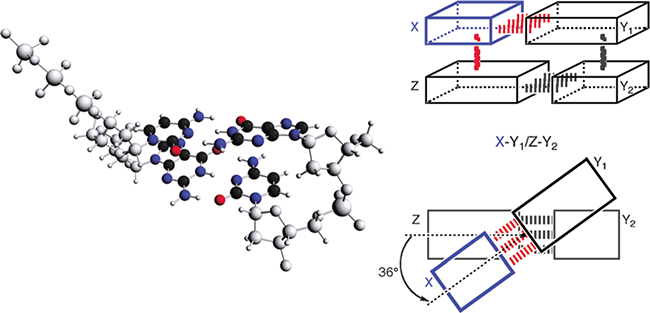
Modeling the hydrogen bonds, pi-stacking and solvent effects in DNA - Software for Chemistry & Materials Software for Chemistry & Materials
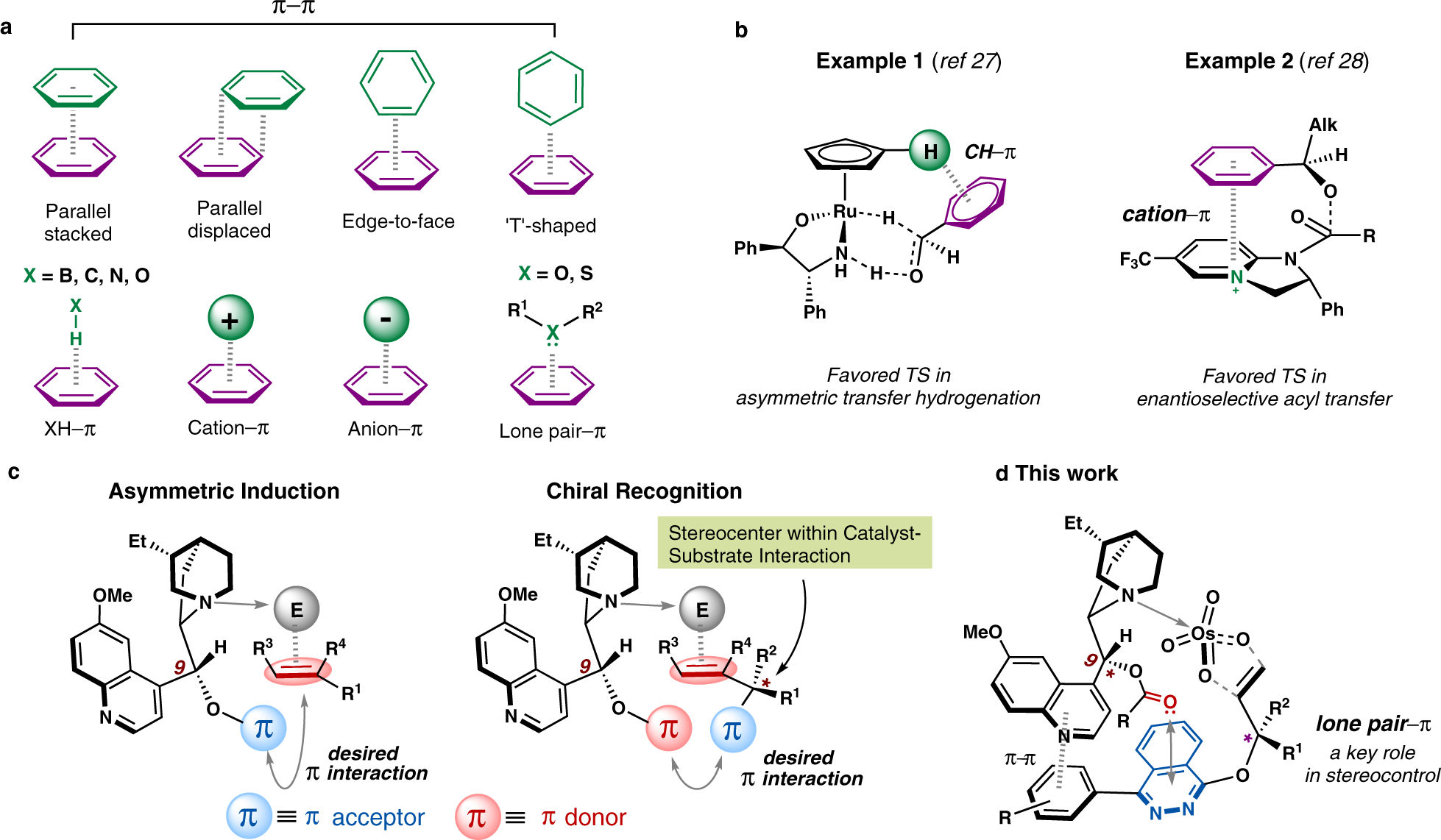
Engineered non-covalent π interactions as key elements for chiral recognition | Nature Communications

π–π stacking face-to-face (left), slipped (middle), and C–H···π (right)... | Download Scientific Diagram


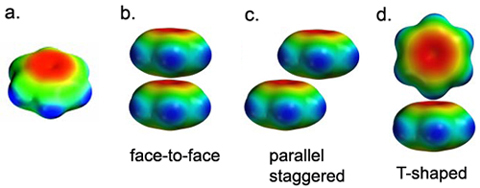




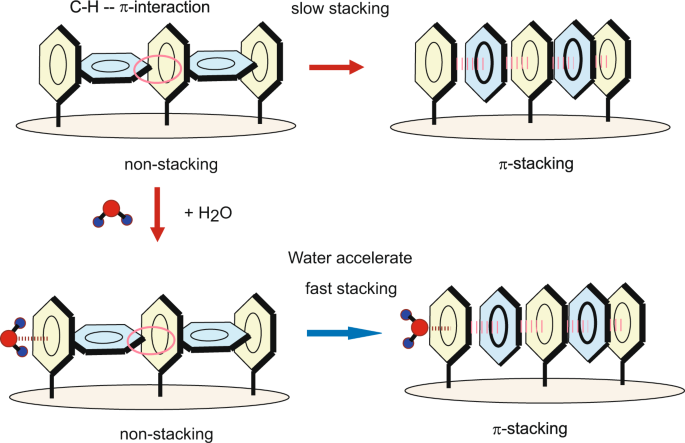
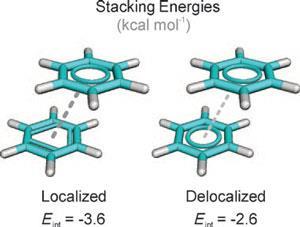
![PDF] Rethinking the term “pi-stacking” | Semantic Scholar PDF] Rethinking the term “pi-stacking” | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/31405337bcf2bd8d49667f6cb9b59793795b4f8d/4-Figure2-1.png)

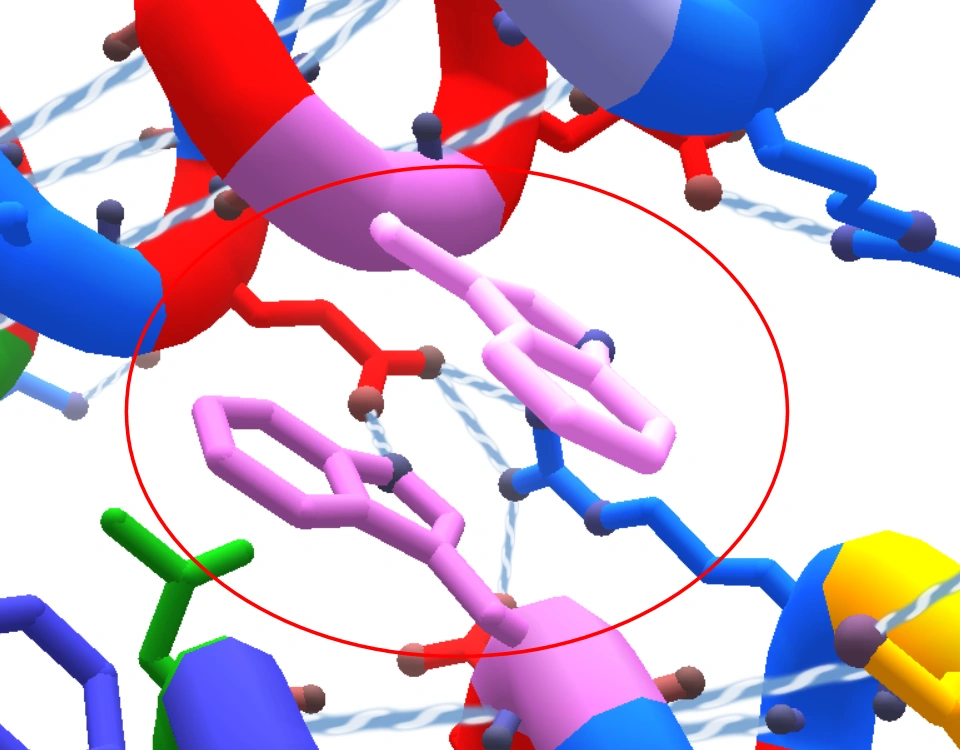
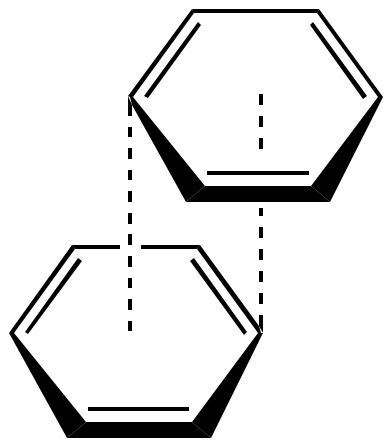

![PDF] Rethinking the term “pi-stacking” | Semantic Scholar PDF] Rethinking the term “pi-stacking” | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/31405337bcf2bd8d49667f6cb9b59793795b4f8d/3-Figure1-1.png)