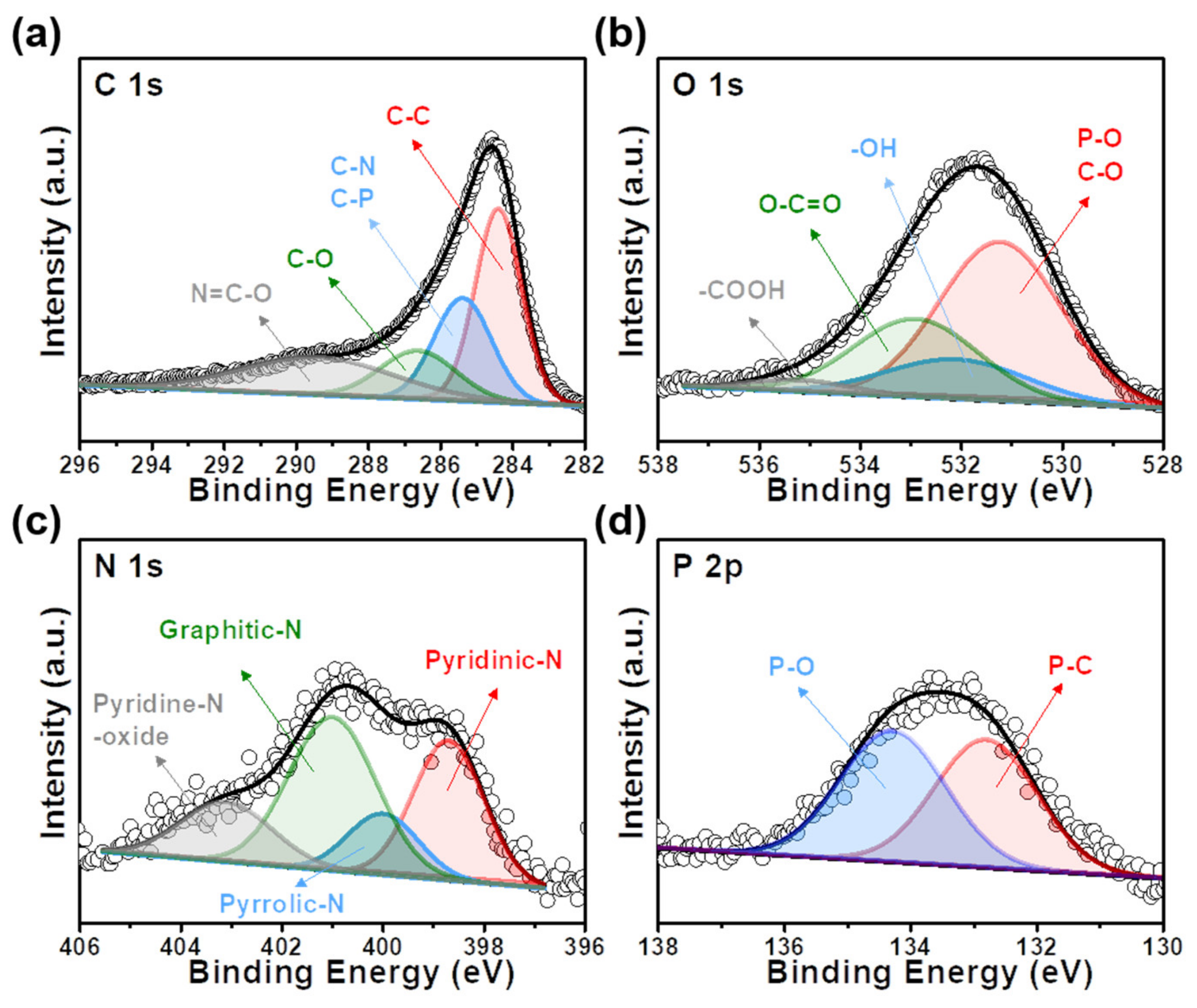
Catalysts | Free Full-Text | Accelerating the Oxygen Reduction Reaction and Oxygen Evolution Reaction Activities of N and P Co-Doped Porous Activated Carbon for Li-O2 Batteries

SOLVED: 1 mol of hydrocarbon P (contains C and H only) required 240 g of oxygen (O2) for complete combustion at standard state heat of combustion of P(l) is -3350 kJ/mol while

Oxygen nonstoichiometry δ dependence on p O2 in Pr x Ce 1-x O 2-δ . The... | Download Scientific Diagram
Equilibrium partial pressure of oxygen p(O2) for selected metal/oxide... | Download Scientific Diagram

Diagram of the chemical potential of oxygen (? P O2 )-temperature (T, K) at | Download Scientific Diagram

4GB 5G O2 sim only for £6 p/m, unlimited mins & texts (12 month - £72 via Moneysupermarket) @ O2 | hotukdeals

Olens | O2 EDITION 1 DAY DISPOSABLE CONTACT LENS (30PCS) (P -1.00) | power : -1.00 | HKTVmall The Largest HK Shopping Platform

Two substance, P4 and O2 are allowed to react completely to form mixture of P4O6 and P4O10 leaving none of the reactants. The composition of final mixture if P4 and O2 is