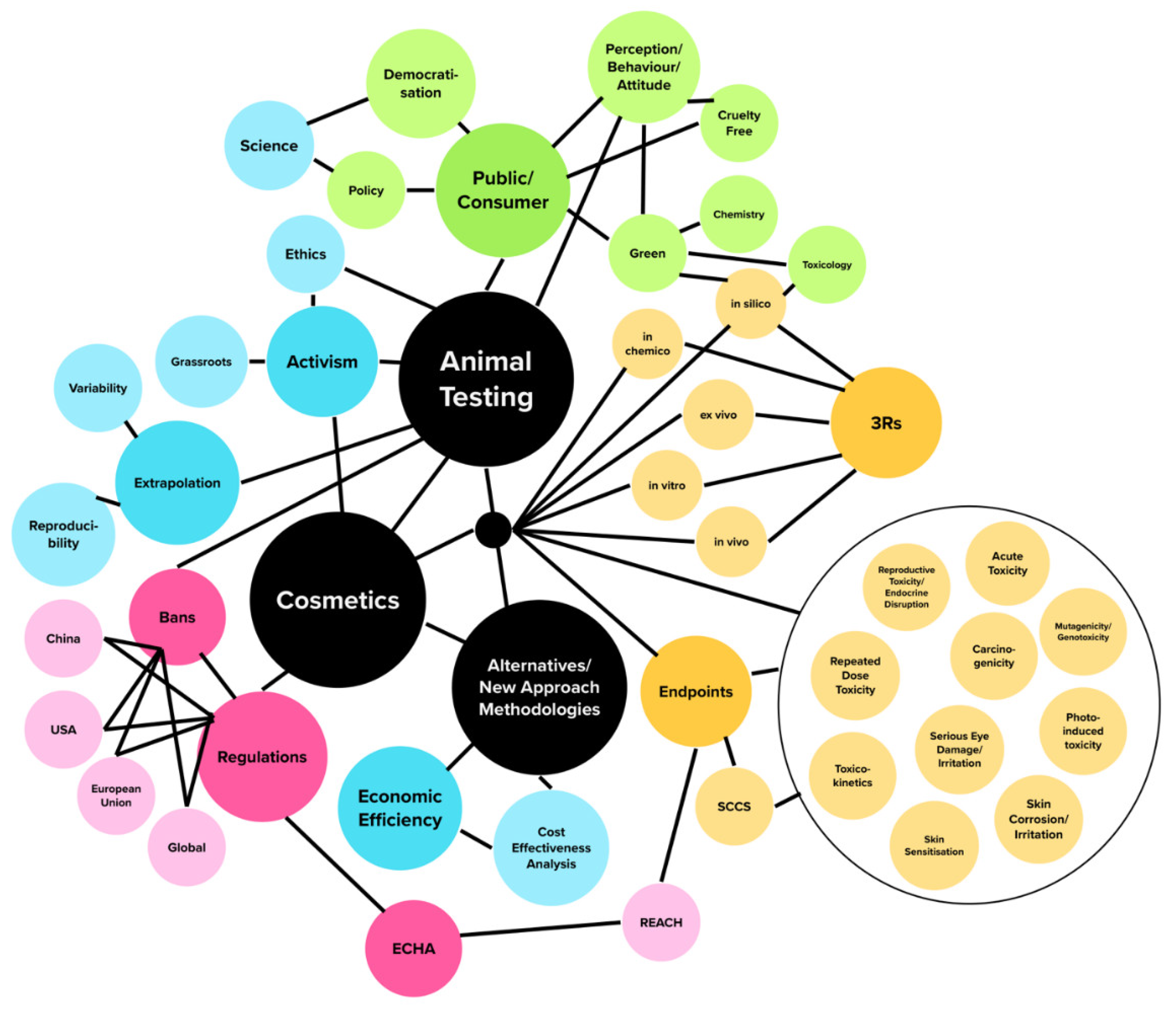
Cosmetics | Free Full-Text | A State-of-the-Art Review on the Alternatives to Animal Testing for the Safety Assessment of Cosmetics
CONFIDENTIAL 1 (2) Helsinki, 18/05/2020 Applicant Nedstar BV Nicolaas Beetsstraat 114H 1054XT Amsterdam Netherlands Sent via R4B

Preliminary draft guidance on the data requirements and assessment of applications for renewal of approval of active substances under BPR March 2019 Hugues. - ppt download
CONFIDENTIAL 1 (2) Helsinki, 04/01/2016 Envirolyte Industries International Ltd Rävala pst 8-C603 10143 Tallin Estonia Sent via