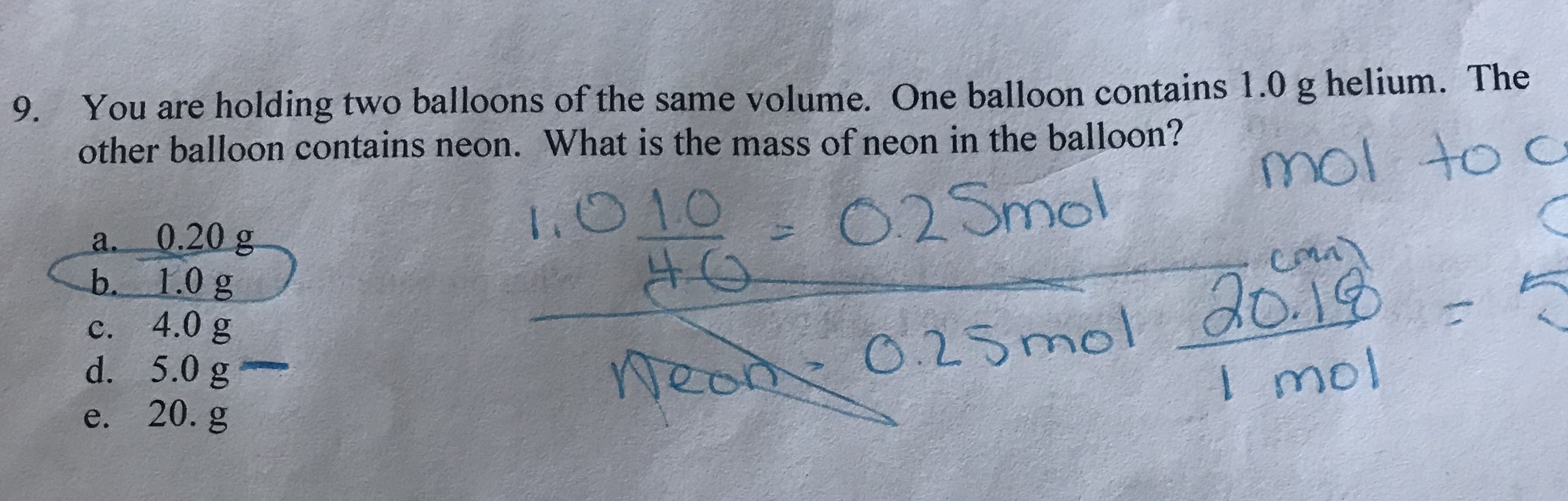Amazon.com: Authentic Models, Floating The Skies Air Balloon, Hanging Home Decor - 22 Inch Height, Historic Hot Air Balloon Model for Home Decor, Detailed Vintage Decorations to Hang from Ceiling - USA :

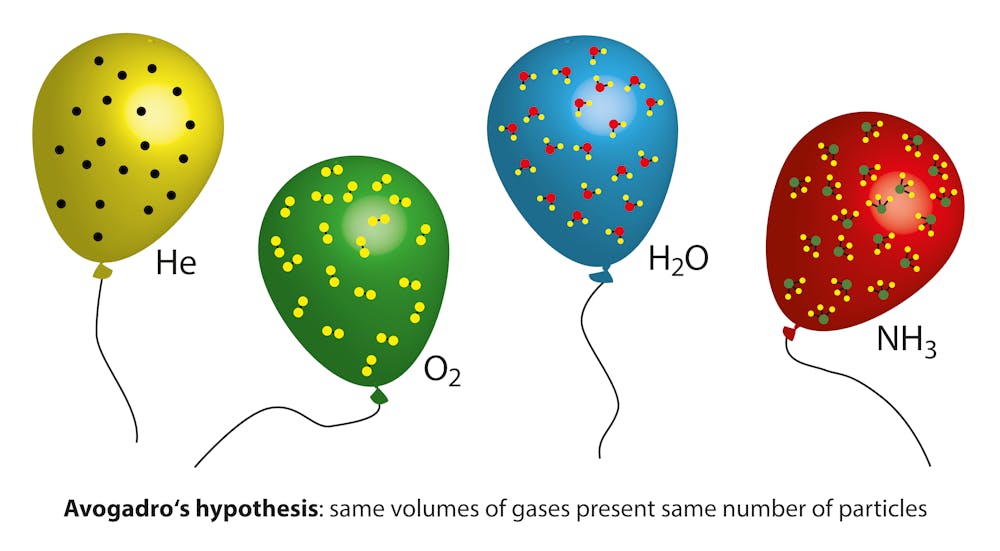
Expanding on Self-Inflating Balloons: Activities Involving Moles, Gas Laws, and Thermochemistry | Chemical Education Xchange

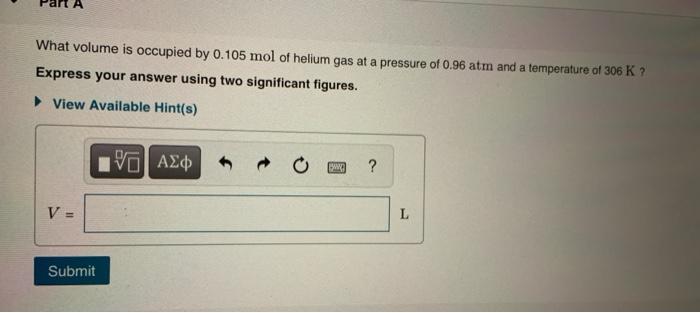
Two moles of ideal helium gas are in a rubber balloon at `30^C.` The balloon is fully expandable... - YouTube
%20(250x141).jpg)
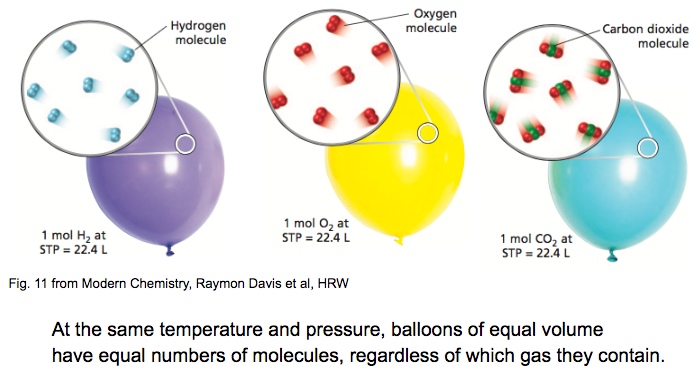
Molecular Structure 9.3-Lecture Demonstrations-Department of Chemistry-University of Massachusetts Amherst

A balloon filled with 0.25 mol of He(g) at 273 K and 1 atm is allowed to rise through the atmosphere. - Brainly.com

SOLVED: A balloon originally has 0.10 moles of helium and has a volume of 0.50 L. If 0.67 grams of He are added to the balloon, what will the new volume be,