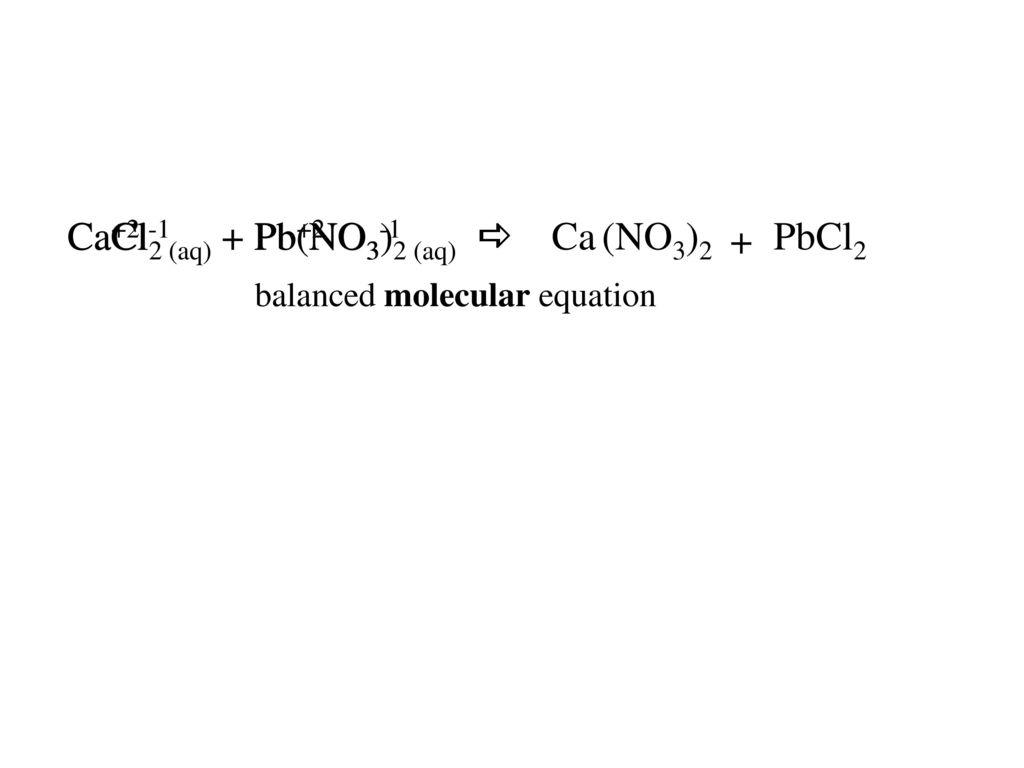
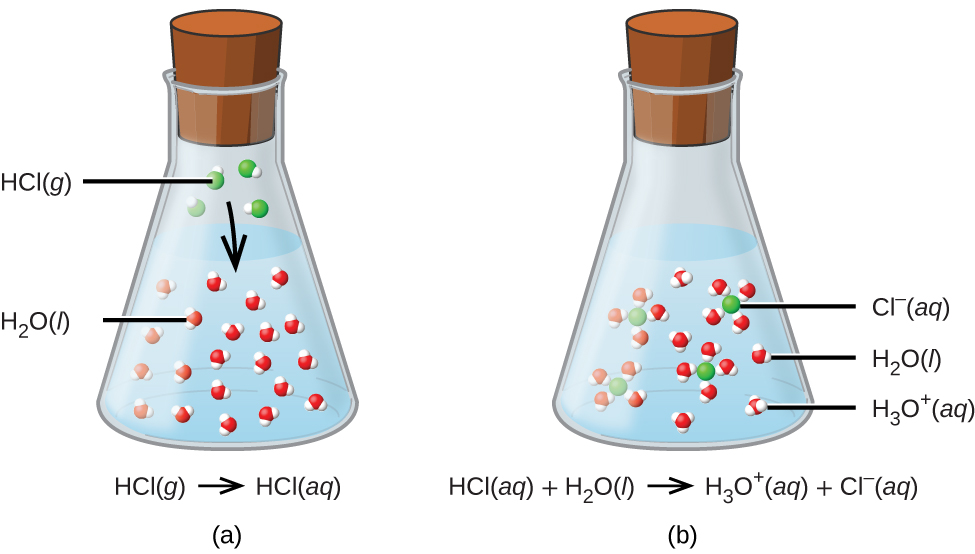
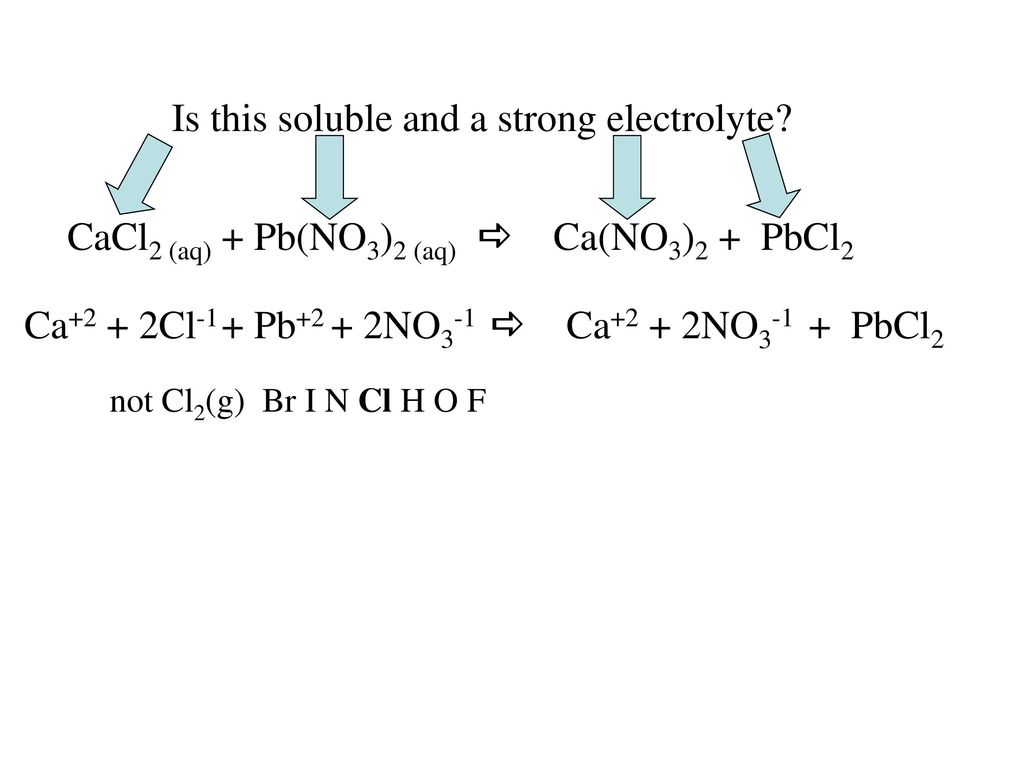![SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ](https://cdn.numerade.com/ask_images/b6e72b4272b24ff5a54297778f90c0b4.jpg)
SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ

Spontaneous exciton dissociation in organic photocatalyst under ambient conditions for highly efficient synthesis of hydrogen peroxide | PNAS

SOLVED: What is the net ionic equation for the following reaction? Al(s) + HCl(aq)- A Al(s) + 2Ht (aq) A1+ (aq) + H2(g) B.2 Al(s) + 6H t (aq) A13+ (aq) +

SOLVED: Example: The following unbalanced net ionic equation is given: AI(s) + Cu2+(aq) A/3+(aq) + Cu(s) What is the balanced net ionic equation? a) 2 Al(s) + Cu2+(aq) e b) 2 Al(s) +