
An adiabatic container fitted with a movable adiabatic piston (operating at 1 atm ) is filled with 2 litre of 2 M H2O2 (aq) solution at 300 K. If H2O2 dissociates following

The variation of concentration of 'A' with time in two experiments starting with two different initial concentration of 'A' is given by the following graph. The reaction is represented by A (aq)
ボルボPENTA対応希土類新スターターモーターAQ 175 A AQ 200 A B C D F AQ 205 A AQ 211 A AQ 225 A B C F 10064 ST 64 ST 64 HD 30119 1109488 1998317 :B00FQSK52E:ニュースプリングショップ - 通販 - Yahoo!ショッピング
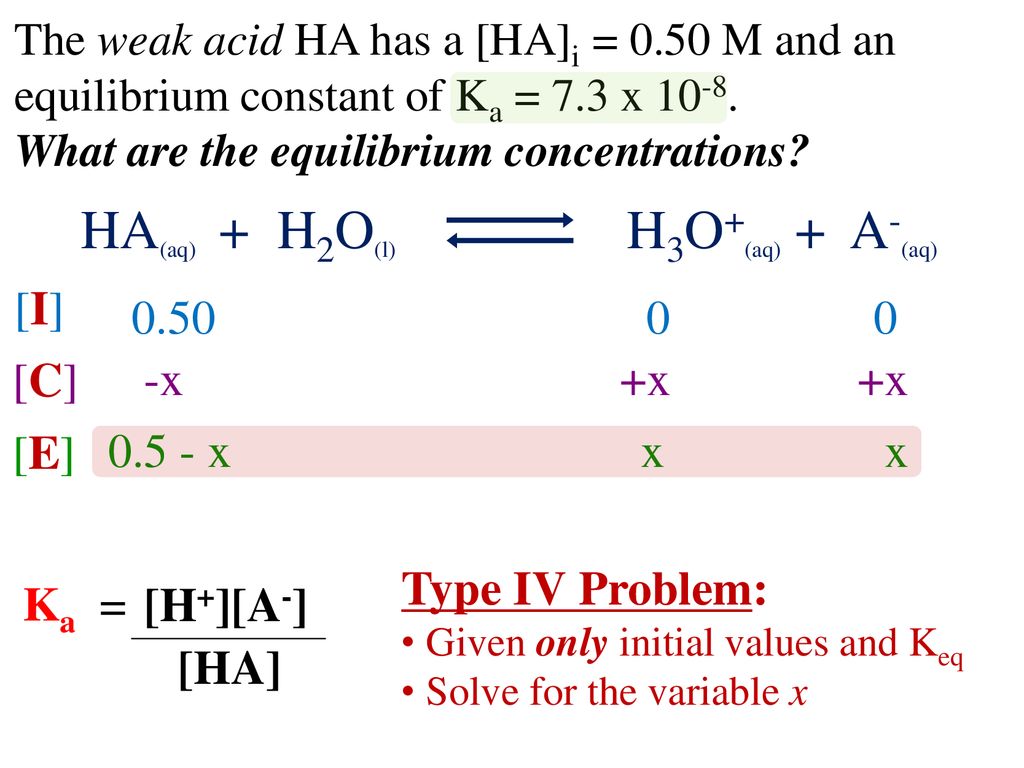
![SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ](https://cdn.numerade.com/ask_previews/be762ce0-8bee-4276-b222-0b2754f62065_large.jpg)












![simple a, aq, aaa, aaq nitials geometric network - Stock Illustration [61606783] - PIXTA simple a, aq, aaa, aaq nitials geometric network - Stock Illustration [61606783] - PIXTA](https://en.pimg.jp/061/606/783/1/61606783.jpg)


![simple a, aq, aaa, aaq nitials geometric network - Stock Illustration [61606789] - PIXTA simple a, aq, aaa, aaq nitials geometric network - Stock Illustration [61606789] - PIXTA](https://en.pimg.jp/061/606/789/1/61606789.jpg)

