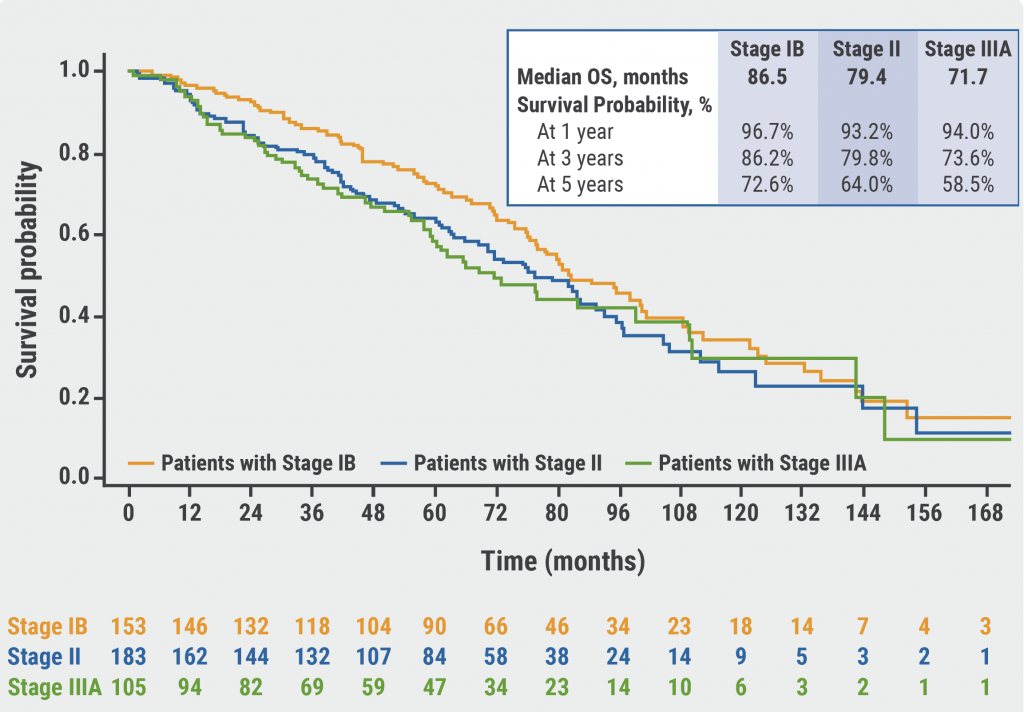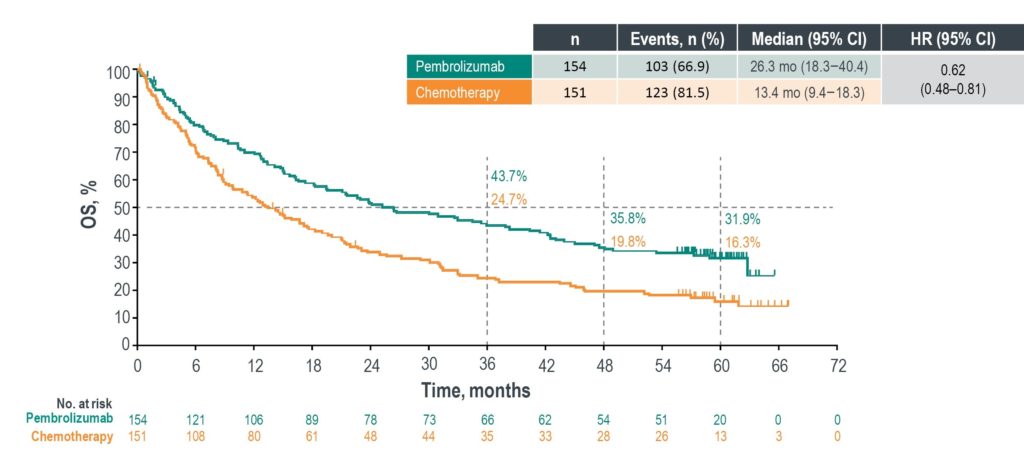
5-year overall survival almost doubled with pembrolizumab for metastatic NSCLC patients with a PD-L1 TPS ≥ 50% and without EGFR/ALK alterations - BJMO
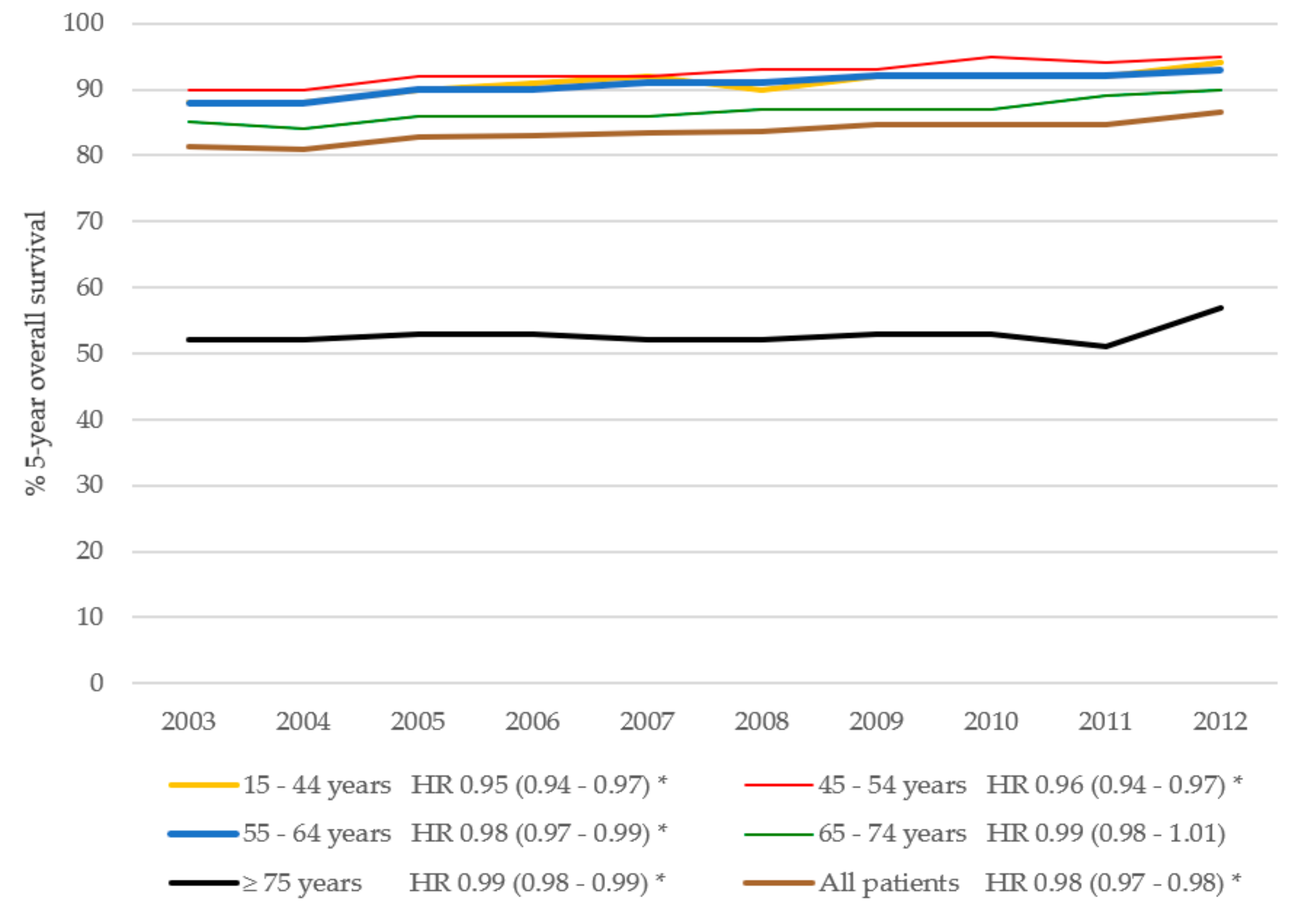
Cancers | Free Full-Text | Trends in Overall Survival and Treatment Patterns in Two Large Population-Based Cohorts of Patients with Breast and Colorectal Cancer

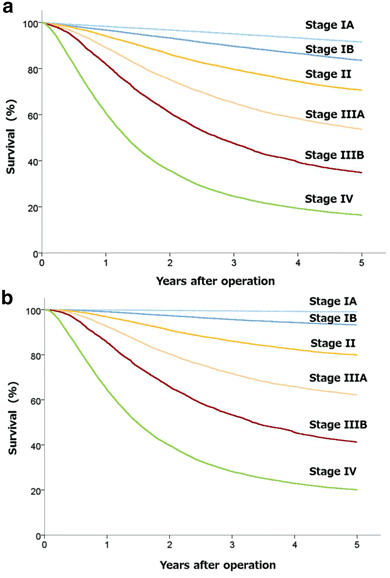
5-year overall survival rates of Different Tumor Stages. After Log-rank... | Download Scientific Diagram

5-year overall survival almost doubled with pembrolizumab for metastatic NSCLC patients with a PD-L1 TPS ≥ 50% and without EGFR/ALK alterations - BJMO
Five-year survival analysis of surgically resected gastric cancer cases in Japan: a retrospective analysis of more than 100,000 patients from the nationwide registry of the Japanese Gastric Cancer Association (2001–2007) | SpringerLink

4baseCare on Twitter: "Research data suggests that 5-year survival rates in different subtypes of Cholangiocarcinoma vary in different stages of the disease. LINK: https://t.co/yh5Cbkcr1j #MedTwitter https://t.co/nhmwIeZAg9" / Twitter

5-year overall survival in patients with lung cancer eligible or ineligible for screening according to US Preventive Services Task Force criteria: a prospective, observational cohort study - The Lancet Oncology

Cancers | Free Full-Text | Trends in Overall Survival and Treatment Patterns in Two Large Population-Based Cohorts of Patients with Breast and Colorectal Cancer

Kaplan-Meier plots of 5-year rates of overall survival in OSCC patients with and without SSPT (a); patients stratified according to the presence of SSPT located at the esophagus or the liver (b),

5-year overall survival in patients with lung cancer eligible or ineligible for screening according to US Preventive Services Task Force criteria: a prospective, observational cohort study - The Lancet Oncology
Significant 5-year overall survival following neoadjuvant nivolumab in lung cancer - Hospital Pharmacy EuropeHospital Pharmacy Europe
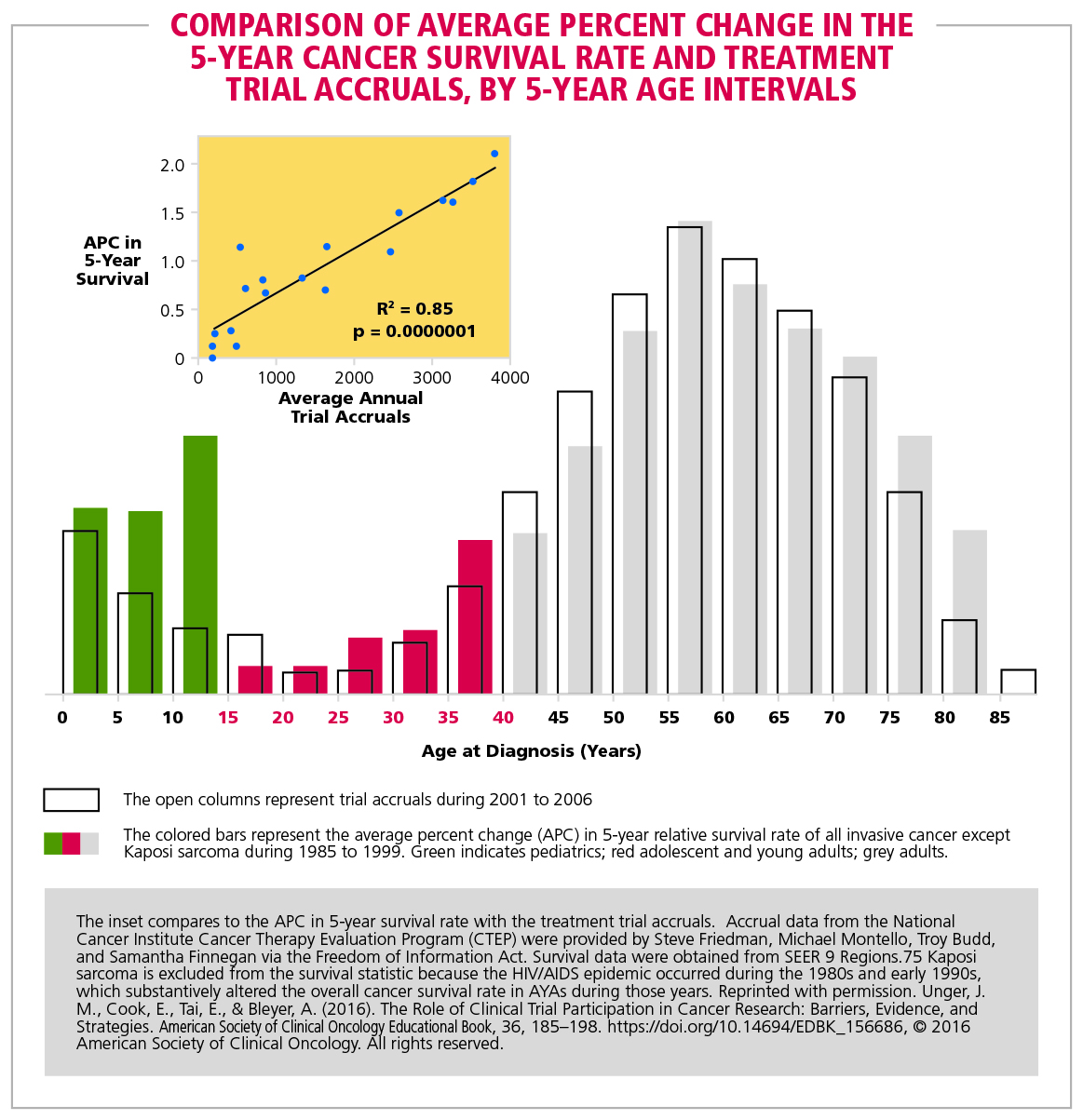
Figure 7. Comparison of average percent change in the 5-year cancer survival rate and treatment trial accruals, by 5-year age intervals | American Cancer Society Cancer Action Network
![PDF] Five-Year Overall Survival for Patients With Advanced Non‒Small-Cell Lung Cancer Treated With Pembrolizumab: Results From the Phase I KEYNOTE-001 Study | Semantic Scholar PDF] Five-Year Overall Survival for Patients With Advanced Non‒Small-Cell Lung Cancer Treated With Pembrolizumab: Results From the Phase I KEYNOTE-001 Study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7cbf64e0a2a5bc6572dc331e433efae660922309/4-Figure1-1.png)