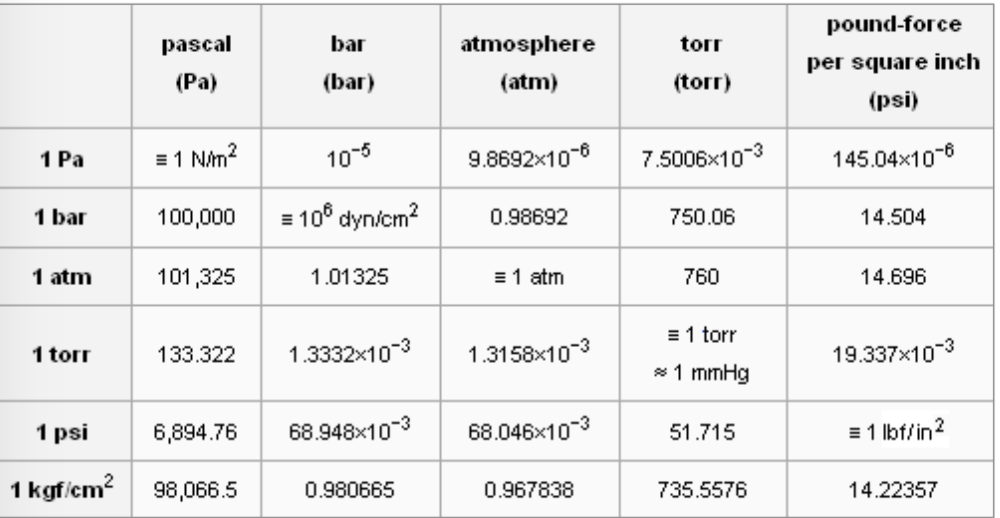
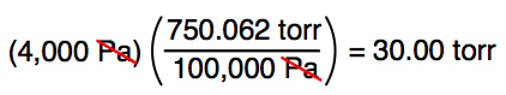Pressure 1 atmosphere ~ 1 bar ~ 760 mm Hg ~ 760 torr ~ 100,000 Pa Ion gauges read in mbar i.e. 1x mbar = 1x atm. Sometimes ion gauges read. - ppt download

SOLUTION: 614 chapter 14 1torr p p gh 250torr 1 06 103 kg m3 9 8m s2 2 0 m brain heart 133 33 pa 94torr b the gauge pressure at the feet of the giraffe is 1tor - Studypool

pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

In an ultrahigh vacuum system (with typical pressure slower than 10^-7 pascal), the pressure is measured to be 1.00 × 10^-10 torr (where 1 torr = 133 Pa). Assuming the temperature is

SOLVED: Convert 661,000 pascal, pa into torr 1 atm 760 torr = 760 mmHg 101.,325 pa 101.325 kpa 1.01325 barr 14.696 p.si 29.921 inHg (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)
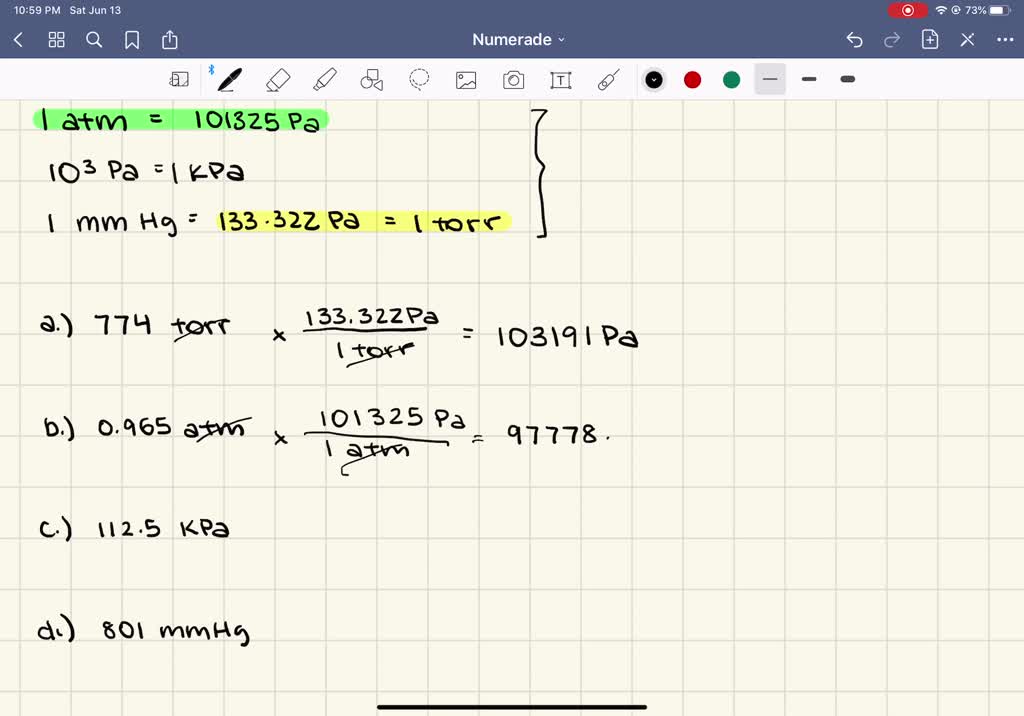
SOLVED:Convert the following pressures into pascals. a. 774 torr b. 0.965 atm c. 112.5 kPa d. 801 mm Hg

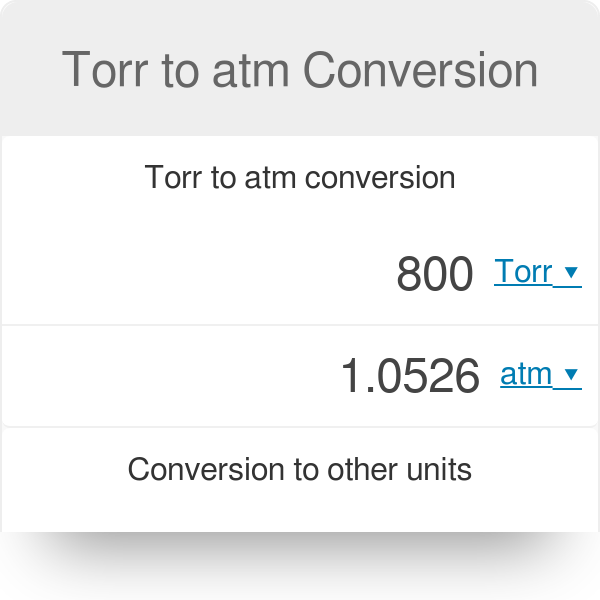
Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

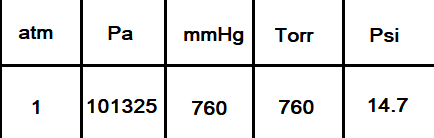
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download